Abstract
The Fanconi anemia (FA) signaling network is critical to maintaining genomic integrity during interphase and mitosis. Biallelic germline mutation of any of the 22 genes that constitute this pathway (FANCA-FANCW) results in the disorder FA, which is characterized by congenital malformations, a predisposition to bone marrow failure, and pediatric acute myeloid leukemias (AMLs). Among the general population, acquired genetic disruptions of the FA pathway are present in 18% of sporadic pediatric AMLs, underscoring the importance of this pathway in the prevention of hematological malignancy. FANCA is the gene most commonly mutated in FA and is one of the most frequently disrupted FA pathway genes in sporadic AML. Therefore, the identification of precision therapies for FANCA-deficient AML is a high clinical priority. To identify synthetic lethal targets of FANCA, we previously identified mitotic phosphosignaling pathways required for the survival of FANCA-/- patient-derived fibroblasts through a kinome-wide shRNA screen. This screen identified the mitotic kinases CHEK1, PLK1, and BUB1 as hits. These findings corroborate work by others who have identified synthetic lethal interactions between PLK1 and the FA pathway components FANCG and BRCA1, suggesting inactivation of the FA pathway may sensitize cancers to PLK1 inhibition. The clinical success of PLK1 inhibitors, such as volasertib and onvansertib, has been limited by the absence of predictive biomarkers of response and adverse side effects.
To test our hypothesis that FA gene mutations may predict sensitivity to PLK1 inhibition we generated an isogenic panel of human AML cells with a stable knockout of the FANCA gene. Utilizing in vitro cell viability assays, we have demonstrated the selective killing of FA-deficient AMLs by pharmacological PLK1 inhibition through low doses of volasertib and onvansertib. We also found that PLK1-inhibition impairs the hematopoietic potential of Fanca-/- murine hematopoietic stem and progenitor cells by performing in vitro bone marrow colony formation assays. Similarly, we demonstrated that primary human AMLs with multiple disruptions within the FA-pathway were hypersensitive to low doses of volasertib.
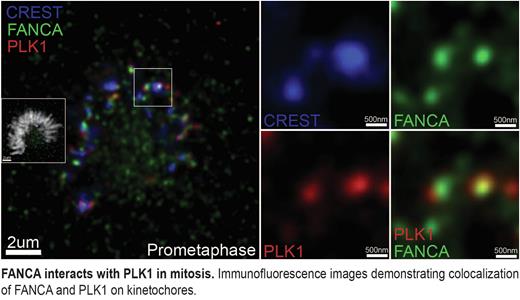
To investigate the mechanism of synthetic lethality, we performed quantitative proteomic analysis of FANCA immunoprecipitates, which identified PLK1 as a novel FANCA-binding partner during mitosis. We validated this finding through high-resolution immunofluorescence microscopy and proximity ligation assays, which revealed a direct interaction between FANCA and PLK1 at kinetochores. Additionally, we demonstrated that volasertib treatment induces monoubiquitination of FANCD2 and colocalization of FANCA with FANCD2 at gamma-H2AX foci. Together, these findings indicate that PLK1 inhibition causes damage to mitotic chromosomes and subsequent activation of the FA pathway. Our work identifies a mitosis-specific vulnerability of FA-deficient cells and suggests that genetic disruptions of the FA pathway may be predictive of sensitivity to PLK1 inhibition, providing a preclinical rationale for the development of precision therapies.
Disclosures
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal